- Buffer and cell culture media
- Bulk product collections and storage
- Chromatography and filtration buffer

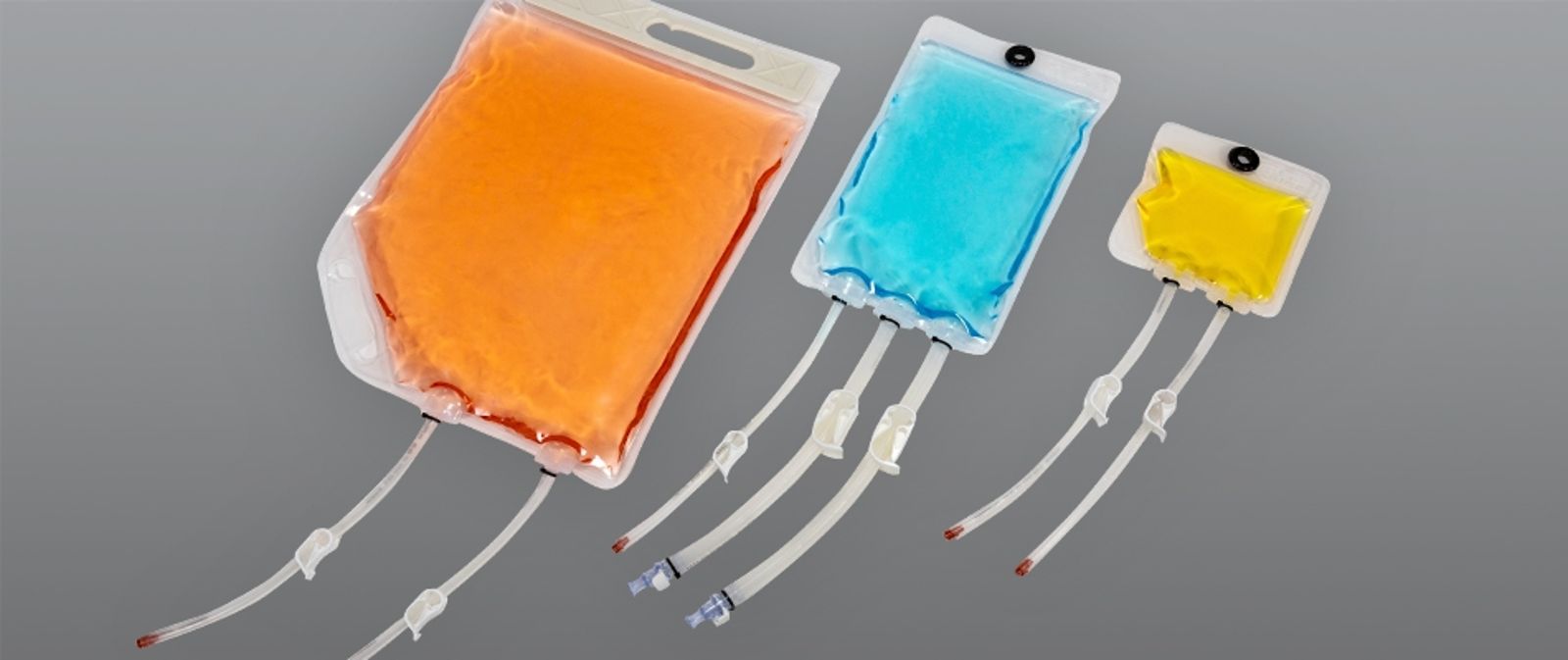
Single-use containers for fluid storage and transfer in cGMP
ILC Dover’s Liquid Bags are single-use fluid storage and transfer bags for handling sterile liquids in the biotherapeutics and cell and gene therapy markets. They’re highly configurable with various sizes, ports, and components to safely store and transfer buffers, media, and bulk cGMP products.
Liquid single-use chambers from ILC Dover are low-profile containers engineered specifically for use in biopharmaceutical environments. Each chamber features careful and intentional design choices, such as edged corners and patented TruFLO ports, in order to maximize product recovery. We designed these bags with availability in mind. All components—film, tubing, and end treatments—can be readily sourced to accelerate lead times.
- Buffer and cell culture media
- Bulk product collections and storage
- Chromatography and filtration buffer

- Fraction collection
- Product sampling and transport

Flexibility
We offer total freedom of customization for all liquid handling designs due to our vendor-agnostic supply chain agreements, so we can source any parts from any vendor for all bioprocess needs.
Integration
Because of our integrated manufacturing suite, ILC Dover's liquid handling solutions are designed, produced, and assembled internally, eliminating the need for numerous vendor agreements and sources.
Support
ILC Dover's dedicated technical staff ensure that your assemblies are optimized for both performance and price, and they're available for support both during the design process and after delivery.
Customization Available
Liquid single-use bioprocessing bags can be engineered to your unique application, with the ability to customize a variety of tubing and connectors, chamber and port sizes, and port configurations.
The containment portion of ILC Dover's liquid bags is made from Renolit 9101 film, a medical-grade polyethylene-based material with multiple low permeability layers. These layers consist of a low-density polyethylene (LDPE) exterior, an ethylene-vinyl-alcohol (EVOH) intermediate oxygen barrier, an ultra-low-density polyethylene (ULDPE) contact interior, and tie layers in between the different polymeric materials.
Renolit 9101 film is designed specifically for use in biopharmaceutical processes and passes a plethora of pharmacopeia compliance standards such as USP Class VI, FDA, ISO, EU, etc. More information on specific compliance codes can be found in the Regulatory Information section included in ILC Dover's validation guide. The film is also free of animal- derived componentry (ADCF) and is manufactured in ISO Class 7 cleanrooms for lot-traceable distribution in global industries.